Advertisements
Advertisements
Question
Explain the mechanism of cleansing action of soaps and detergents.
Solution
1. An example of soap is sodium palmitate.
2. The cleansing action of soap is directly related to the structure of carboxylate ions (palmitate ions) present in the soap.
3. The structure of palmitate exhibits dual polarity
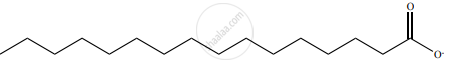
4. The hydrocarbon portion is non polar, hydrophobic and soluble in oils and greases but not in water.
5. Carboxyl portion is polar, hydrophilic, and soluble in water.
6. Dirt in the cloth is due to the presence of dust particles intact or grease which sticks.
7. When soap is added to an oily or greasy part of the cloth, the hydrocarbon part of the soap dissolves in the grease, leaving the negatively charged carboxylate end exposed to the grease surface.
8. At the same time the negatively charged carboxylate groups are strongly attracted by water, forming small droplets called micelles, and grease is floated away from the solid object.
9. When clothes are rinsed with water, the grease goes with them.
10. As a result, the doth gets free from dirt and the droplets are washed away with water.
11. The micelles do not combine into large drops because their surfaces are all negatively charged and repel each other.
12. The cleansing action of soap depends upon its tendency to act as an emulsifying agent between water and water-insoluble greases.
13. The cleansing action of detergents is similar to the cleansing action of soaps.
