Advertisements
Advertisements
Question
Explain the origin of paramagnetism on the basis of atomic structure.
Advertisements
Solution
The presence of persistent atomic or molecular magnetic dipole moments is necessary for paramagnetism. The spin and orbital magnetic moments of its electrons combine in a specific way to produce the intrinsic net atomic magnetic moment.
The internal magnetic field produced by the magnetic moments of neighbouring electrons has an impact on the spin magnetic moments of the electrons in matter. The spin magnetic moments process the field direction due to this internal field, which ranges from ∼10−2 T to 10−1 T. The internal field is constantly fluctuating at room temperature due to the thermal mobility of the electrons, resulting in random orientations for the spin magnetic moments (Fig. (a). Therefore, a paramagnetic substance is not magnetized in the absence of an external magnetizing field.
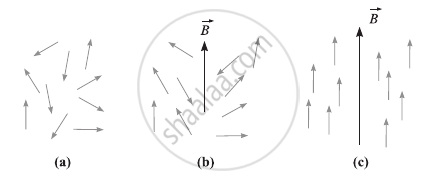
Magnetic dipole moments in a paramagnetic sample (a) randomly directed in the absence of a magnetizing field (b) partial alignment on the application of an external field (c) aligned to saturation at a very low temperature or strong field.
The spin magnetic moments tend to align parallel to the direction of the external field when the applied field intensity is higher than the internal field. However, full alignment is impeded by the randomizing impact of heat agitation, as shown in Fig. (b). As a result, a paramagnetic material is only weakly magnetized in the direction of the magnetizing field at room temperature when it is exposed to a magnetic field.
The magnetic dipole moments are efficiently oriented parallel to the field to have the least magnetic potential energy and the magnetization reaches saturation if the external field is sufficiently strong or the temperature is very low, as seen in Fig. (c).
