Advertisements
Advertisements
Question
Explain the term ‘Interconversion of matter’. With reference to ice, water, and water vapour show diagrammatically the change of state of matter from solid to liquid to gaseous and back to the original state.
Solution
INTERCONVERSION OF MATTER:
“Change of state of matter from one state to another state and back to its original state is called inter-conversion of matter.”
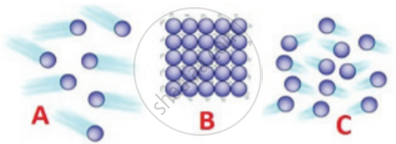
The diagram showing the Change of State of Matter:

(a) ICE TO WATER (MELTING): Conversion of a solid into a liquid on heating
e.g. ice to water.
(b) WATER TO WATER VAPOUR (VAPORISATION): Conversion of a – liquid into vapours (or gas) on heating
e.g. water to water vapour.
(c) WATER VAPOUR TO WATER (LIQUEFACTION or CONDENSATION): Conversion of VAPOUR (or gas) into a LIQUID on cooling
e.g. water vapour to water.
(d) WATER TO ICE (SOLIDIFICATION OR FREEZING): Conversion of a LIQUID into a SOLID
e.g. water to ice.
RELATED QUESTIONS
"A substance has a definite shape as well as a definite volume." Which physical state is represented by this statement?
State two characteristic properties of a solid.
Arrange the following substances in increasing order of force of attraction between their particles (keeping the substance having the minimum force of attraction first) : Water, Sugar, Oxygen
Give reasons
We can walk through air.
Write whether the following statements are true or false:
Only water can exist in three different states
Describe simple experiments to prove that gases occupy space.
State the property of the following substance.
Magnesium reacts with dilute hydrochloric acid, liberating hydrogen gas.
Identify the state of matter based on the arrangement of the molecules.
How the matter is grouped on the basis of physical states?
Give two examples of diffusion.
