Advertisements
Advertisements
Question
Find out the reaction intermediate and molecularity of the following reactions.
\[\ce{NO_{(g)} + Cl2_{(g)} -> NOCl_{2(g)}}\]
\[\ce{NOCl_{2(g)} + NO_{(g)} -> 2NOCl_{(g)}}\]
Draw the structure of BrF5 and HOCl.
Short Note
Solution
Reaction intermediate = NOC12(g)
Molecularity of the reaction = 2
Structure of:
| Bromine pentafluoride (BrF5) | Hypochlorous acid (HOCl) |
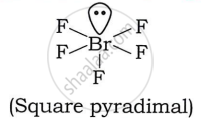 |
\[\begin{array}{cc} \ce{O}\\ \phantom{}/\phantom{...}\backslash\\ \ce{\underset{(Angular or 'V' shaped)}{H\phantom{....}Cl}} \end{array}\] |
shaalaa.com
Molecularity of Elementary Reactions
Is there an error in this question or solution?
