Advertisements
Advertisements
Question
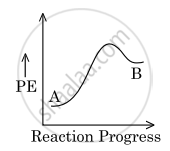
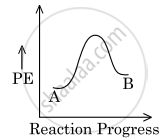
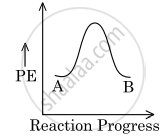
For a reaction, A `→` B, the enthalpy of reaction is −4.2 kJ mol−1 and the enthalpy of activation is 9.6 kJ mol−1. The correct potential energy profile for the reaction is shown in the option.
Options
MCQ
Solution
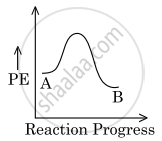
shaalaa.com
Enthalpy Change, ∆_rH of a Reaction - Reaction Enthalpy - Standard Enthalpy of Reactions
Is there an error in this question or solution?