Advertisements
Advertisements
Question
For the reaction, \[\ce{2Cl_{(g)} → Cl_{2(g)}}\], what are the signs of ΔH and ΔS?
Solution 1
ΔH and ΔS are negative
The given reaction represents the formation of chlorine molecule from chlorine atoms. Here, bond formation is taking place. Therefore, energy is being released. Hence, ΔH is negative.
Also, two moles of atoms have more randomness than one mole of a molecule. Since spontaneity is decreased, ΔS is negative for the given reaction.
Solution 2
∆H : negative (- ve) because energy is released in bond formation
∆S : negative (- ve) because entropy decreases when atoms combine to form molecules.
APPEARS IN
RELATED QUESTIONS
A reaction, A + B → C + D + q is found to have a positive entropy change. The reaction will be ______.
The entropy change can be calculated by using the expression ∆S = `q_(rev)/T`. When water freezes in a glass beaker, choose the correct statement amongst the following :
The spontaneity means, having the potential to proceed without the assistance of external agency. The processes which occur spontaneously are:
(i) Flow of heat from colder to warmer body.
(ii) Gas in a container contracting into one corner.
(iii) Gas expanding to fill the available volume.
(iv) Burning carbon in oxygen to give carbon dioxide.
Enthalpy diagram for a particular reaction is given in figure. Is it possible to decide spontaneity of a reaction from given diagram. Explain.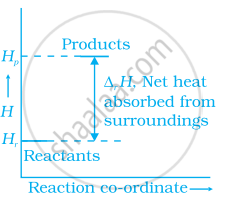
Match the following processes with entropy change:
| Reaction | Entropy change |
| (i) A liquid vapourises | (a) ∆S = 0 |
| (ii) Reaction is non-spontaneous at all temperatures and ∆H is positive |
(b) ∆S = positive |
| (iii) Reversible expansion of an ideal gas | (c) ∆S = negative |
Assertion (A): A liquid crystallises into a solid and is accompanied by decrease in entropy.
Reason (R): In crystals, molecules organise in an ordered manner.
For a sample of perfect gas when its pressure is changed isothermally from Pi to Pf, the entropy change is given by ______.
For the irreversible expansion of an ideal gas under isothermal conditions, the correct option is ______.
The process with negative entropy change is ______.
A reaction at 1 bar is non-spontaneous at low temperatures but becomes spontaneous at high temperatures. Identify the correct statement about the reaction among the following.
