Advertisements
Advertisements
Question
Give a reason for the following statement:
Components in a mixture are present in varying proportions and not in a fixed proportion.
Solution
A mixture is an impure substance containing more than one element or compound, mechanically mixed together in any proportion and the mixture retains the properties of its constituents i.e. elements or compounds.
APPEARS IN
RELATED QUESTIONS
What is the difference between a pure substance and a mixture ?
Define: Compounds
Write the Chemical Name of the Following and Also Give Their Molecular Formulae:
Sand
Name: a Liquid Metal
Give two examples of the following.
Elements which are liquids
Define: Molecule
Name: A form of carbon used as a gem
Fill in the blank
A metal which is a liquid at room temperature is _______.
Indicate whether the following statement is true or false:
Molecules combine to form atoms
Differentiate between the terms –
elements, compounds & mixtures.
Give the name of the radical - CO3
Complete the statement given below by filling in the blank with the correct word.
Element ______ has the symbol derived from its Latin name ‘Argentum’
Select the correct answer from the choice given below.
The unreactive non-metallic element
Select the correct answer from the choice given below.
The element which is malleable
Define the term ‘compound’. In the compound carbon dioxide – the elements carbon & oxygen are combined in a fixed ratio. Explain.
Select the correct answer from A, B, C, D & E for the statement given below :
A metalloid.
Match the separation of components in List I with the most appropriate process in List II.
| List I | List II |
| 1. Naphthalene from naphthalene & sodium chloride. | A: Separating funnel |
| 2. Cream from milk. | B: Sublimation |
| 3. Kerosene oil from kerosene oil & water. | C: Boiling |
| 4. Lead nitrate from an aqueous solution of lead nitrate. | D: Centrifugation |
| 5. Ammonia from an aqueous solution of ammonia. | E: Distillation |
Match the following:
| 1. | Co2 | a. | Robert Boyle |
| 2. | Sulfur | b. | Making mobile phones |
| 3. | Element | c. | triatomic molecule |
| 4. | Gallium | d. | Polyatomic molecule |
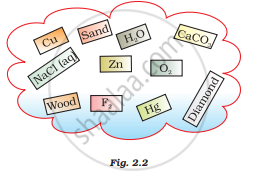
Classify the substances given in Fig. 2.2 into elements and compounds