Advertisements
Advertisements
Question
Give a valence bond description of the bonding in the high spin complex [MnCl6]3− Include orbital diagram for the free metal ion and the metal ion in the complex. Indicate which hybrid orbitals the metal ion uses for bonding. Specify the number of unpaired electrons.
Answer in Brief
Solution
[MnCl6]3− (High spin)
- Oxidation state of manganese = + 3, Mn3+
Electron configuration of Mn3+ : [Ar] 3d4
Orbital diagram of free metal ion: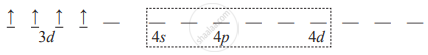
- Because the complex is high spin, pairing of electrons will not occur. So in the complex orbital diagram of Mn3+ will remain the same.
∴ d 2sp3 hybridization is not possible. Hence, one 4s, three 4p and two 4d orbitals hybridize to give six sp3d 2 vacant hybrid orbitals. - The six vacant hybrid orbitals of Mn3+ overlap with six filled orbitals of six ligands forming Mn → Cl coordinate bonds.

- There are 4 unpaired electrons in the complex
shaalaa.com
Theories of Bonding in Complexes
Is there an error in this question or solution?
