Advertisements
Advertisements
Question
Give the inter-relationship of methane, methyl alcohol, formaldehyde and formic acid with conditions
Solution
(1) Methane to methyl alcohol:
When a mixture of methane and oxygen is compressed to about 120atm pressure and passed over copper tubes at 475K, ethyl alcohol is formed.
`2CH_4 + O_2` 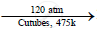 `2CH_3OOH`
`2CH_3OOH`
(b) Methane to formaldehyde:
When mixture of methane and oxygen is passed through heated molybdenum oxide, the mixture is oxidized to Formaldehyde.
`CH_4 + O_2 ` 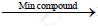 `2HCOOH + 2H_2O`
`2HCOOH + 2H_2O`
(c) Methane to Formic acid:
When a manganese based catalyst is used methane is oxidized to formic acid.
`2CH_4 + 3O_2` 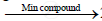 `2HCOOH + 2H_2O`
`2HCOOH + 2H_2O`
shaalaa.com
Hydrocarbons: Alkenes
Is there an error in this question or solution?
