Advertisements
Advertisements
Question
Give VBT description of the bonding in a square planar complex [Cu(NH3) 4]2+. Show orbital diagrams for free metal ion and metal ion in the complex. Which hybrid orbitals are used by metal for bonding? State magnetic nature of the complex.
Answer in Brief
Solution
[Cu(NH3)4]2+ (Square planar)
- Oxidation state of copper is +2, Cu2+ Electron configuration of metal ion is [Ar]3d9.
- Orbital diagram of free metal ion:
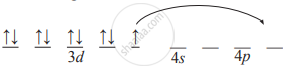
- Because the complex is square planar Cu2+ must be dsp2 hybridized. In order to make one d orbital vacant, unpaired electron of d orbital is transferred to the last 4p orbital. Thus,
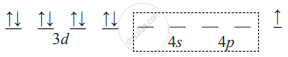
- One 3d orbital, one 4s orbital and two 4p orbitals of Cu2+ ion hybridize to produce four dsp2 vacant hybrid orbitals. These orbitals overlap with filled orbitals of 4 ligand donor atoms.
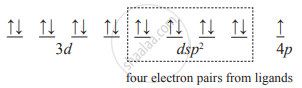
- The complex contains one unpaired electron. Therefore, it is paramagnetic.
shaalaa.com
Theories of Bonding in Complexes
Is there an error in this question or solution?
