Advertisements
Advertisements
Question
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): An ether is more volatile than an alcohol of comparable molecular mass
Reason (R): Ethers are polar in nature.
Select the most appropriate answer from the options given below:
Options
Both A and R are true and R is the correct explanation of A.
Both A and R are true but R is not the correct explanation of A.
A is true but R is false.
A is false but R is true.
Solution
Both A and R are true but R is not the correct explanation of A.
Explanation:
A and R are two different statements about ethers.
The correct reason is that hydrogen bonding does not exist amongst ether molecules.
APPEARS IN
RELATED QUESTIONS
Write the product(s) in the following reactions
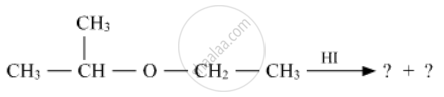
Give simple chemical tests to distinguish between the following pairs of compounds :
Propanol and 2-methylpropan-2-ol
In the following reaction:
\[\ce{C2H5-O-C2H5 + 4[H] ->[Red] 2X + H2O}\] X is:
tert-Butyl methyl ether on heating with HI gives a mixture of ____________.
Which of the following ether has the highest boiling point?
