Advertisements
Advertisements
Question
Gold does not get corroded due to oxidation. Why?
Short Note
Solution
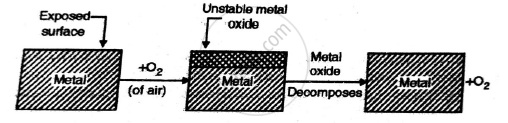
Gold forms an unstable oxide film on metal surface.They get decomposed on metal surfaces back to metal & oxygen.Consequently, oxidation corrosion is not possible such a case.Thus Pt,Au and Ag don’t go under corrosion
Metal+Oxygen →Metal oxide →Metal+Oxygen.
shaalaa.com
Factors Affecting the Rate of Corrosion- Nature of Metal
Is there an error in this question or solution?
