Advertisements
Advertisements
Question
How are the following conversions carried out?
propene to 1-iodopropane
Solution
\[\ce{\underset{\text{Propene}}{H3C - CH = CH2 + HBr}->[Peroxide]\underset{\text{1-Bromopropane}}{H3C - CH2 - CH2Br}}\]
\[\ce{\underset{\text{1-Bromopropane}}{H3C - CH2 - CH2Br + NaI}->[Acetone]\underset{\text{1-Iodopropane}}{CH3CH2CH2I} + NaBr↓}\]
RELATED QUESTIONS
An alkyl chloride on Wurtz reaction gives 2,2,5,5-tetramethylhexane. The same alkyl chloride on reduction with a zinc-copper couple in alcohol gives hydrocarbon with molecular formula \[\ce{C5H12}\]. What is the structure of alkyl chloride?
Write the major product of the following reaction.
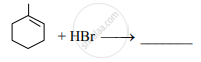
Write IUPAC name of the product ‘B’ in the following reaction sequence.
\[\ce{C2H5OH ->[NaBr/H2SO4][\Delta] A ->[Nal][acetone]B}\]
How are the following conversions carried out?
propene to 2-nitropropane
Write chemical equations indicating the action of following on bromobenzene.
CH3COCl/anhy. AlCl3
Write chemical equations indicating the action of following on bromobenzene.
fuming H2SO4
Write chemical equations indicating the action of following on bromobenzene.
conc. HNO3/conc. H2SO4
An organic compound A with molecular formula C4H10O on treatment with phosphorus pentachloride gives alkyl chloride. Alkyl chloride on treatment with Mg in presence of dry ether gives a highly reactive compound B.
Compound B reacts with water to give hydrocarbon C. Alkyl chloride on treatment with Na in dry ether as a solvent gives alkane, 2,2,3,3-tetramethylbutane. Identify ‘A’, ‘B’, ‘C’
Write the equations for the preparation of ethyl chloride using
- Hydrogen halide
- ethene
- Thionyl chloride
Which of these methods is preferred and why?
Lucas reagent is a mixture of ____________.
Identify 'B' in the following reaction.
\[\ce{C2H6 ->[Br2][AlBr3] A ->[CH3COOAg][\Delta] B}\]
Which of the following reactions does NOT occur?
Which among the following compounds is used to decaffeinate coffee?
Which of the following is NOT an example of freons?
Phosgene is the common name for ______.
End product of the following sequence of reactions is:

Write use of CFC.
Write environmental effect of CFC.
State the environmental effects of the following compound:
Freons (CCl2F2, CCl3F, CHClF2)
