Advertisements
Advertisements
Question
How can you experimentally prove water is a bad conductor of heat? How is it possible to heat water easily while cooking?
Solution
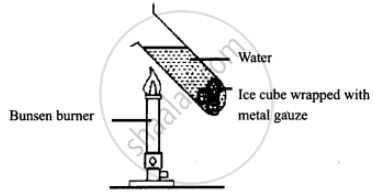
Take a glass tube and drop an ice cube wrapped in wire gauze in it.
Now fill 3/4th of this tube with water and place it above the burner as shown in the figure.
You can observe that the water boils at the edge and the ice present in the bottom of the tube has not melted indicating that heat has not reached the bottom where the ice cube is present. This proves that water is a bad conductor of heat.
It is easy to heat water easily or quickly while cooking. This is because, while cooking the vessel or pan is usually covered with a lid.
This leads to three things;
- Radiation from the hot water is reflected back into the pan rather than being emitted
- Free convection is effectively eliminated, and
- Evaporative cooling’ is also eliminated.
This in turn allows the water to be heated more easily.
APPEARS IN
RELATED QUESTIONS
Match the following.
| Conduction | Liquid |
| Convection | Gas to liquid |
| Radiation | Solid to gas |
| Sublimation | gas |
| Condensation | Solid |
What are the applications of conduction in our daily life?
Two cylindrical rods of same length have the area of cross section in the ratio 2:1. If both the rods are made up of same material, which of them conduct heat faster?
Mud houses are cooler in summer and warmer in winter because ______.
When two objects are in thermal contact, the heat is transferred by ______.
What is the transfer of heat?
Why do we wear woolen clothes in winter?
When a dog keeps out its tongue and breathes hard, the moisture on the tongue turns into ______ and it evaporates.
Name a device that prevents loss of energy (or gain) by conduction, convection, and radiation.
Give some practical applications of conduction in daily life.
