Advertisements
Advertisements
Question
How will you carry out the following conversions?
Ethene to acetic acid
Solution
Ethene to acetic acid
\[\ce{C2H4 + H2 ->[Pt] C2H6}\]
\[\ce{C2h6 + O2 ->[MoO][Conc.H2SO4] CH3CHO + H2O}\]
\[\ce{CH3CHO + [O] ->[K2Cr2O7[Conc.H2SO4] CH3COOH}\]
APPEARS IN
RELATED QUESTIONS
Complete the following chemical equations:
C2H5OH+CH3COOH`("conc."H_2SO_4)/`>
Complete the following chemical equations : C2H5OH + Na →
Three organic compounds A, B and C have the following molecular formulae: C4H10O
Which compound contains a carboxyl group? Write its name and structural formula.
The substance which can produce brisk effervescence with baking soda solution is:
(a) ethanol
(b) vegetable oil
(c) vinegar
(d) soap solution(b) hydrocarbon ends directed towards the centre and ionic ends directed outwards
In a soap micelle, the soap molecules are arranged radially with the hydrocarbon ends, i.e. hydrophobic, directed towards the centre; and, ionic ends, i.e. hydrophilic, directed outwards.
What do you notice when acetic acid reacts with litmus?
What do you observe when acetic acid is added to ethyl alcohol in the presence of sulphuric acid?
A student is studying the properties of acetic acid in his school laboratory. List two physical and two chemical properties which he must observe and note in his record book.
Write the molecular formula of the given compound.
Sodium ethanoate
What are catalysts?
The reaction between acetic acid and sodium carbonate is shown in the following figure.
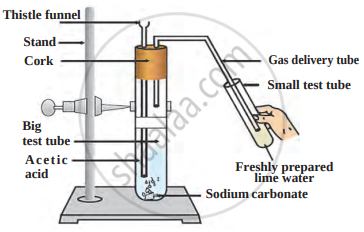
Answer the questions with the help of a diagram.
- Which gas does come out as effervescence in the big test tube?
- What is the colour change in the lime water present in the small test tube?
- Write the related reaction.
