Advertisements
Advertisements
Question
How will you prepare acetamide from methyl cyanide?
Solution
\[\begin{array}{cc}
\ce{\underset{(Methyl cyanide)}{CH3 - C ≡ N} ->[Con. HCl][H2O/OH] CH3 - C - NH2}\\
\phantom{.......................}||\\
\phantom{.......................}\ce{\underset{(Acetamide)}{O}}
\end{array}\]
APPEARS IN
RELATED QUESTIONS
Write reaction showing aldol condensation of cyclohexanone.
Write the name of the product when ketones react with 1,2-diol in presence of dry HCl.
In Clemmensen's reaction, ketone gives ____________.
\[\ce{CH3Br ->[KNC] (A) ->[H3O^+] (B) ->[PCl5] (C)}\] product (C) is:
Identify A, B and C.
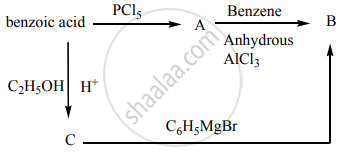
How will you convert benzaldehyde into the following compound?
Benzoic acid
Identify the reaction in which carbonyl group of aldehydes and ketones is reduced to methylene group on treatment with zinc-amalgam and cone. HCI.
What is the action of hydrogen cyanide on the following?
Acetone
Identify B in the following reaction:
\[\begin{array}{cc}
\ce{O}\phantom{........}\\
||\phantom{........}\\
\ce{\underset{(Acetone)}{CH3 - C - CH3}->[CrO3] B}
\end{array}\]
Write the structure of the major product of the aldol condensation of benzaldehyde with acetone.
