Advertisements
Advertisements
Question
How would you prepare:
sodium hydrogen carbonate crystals.
Answer in Brief
Solution
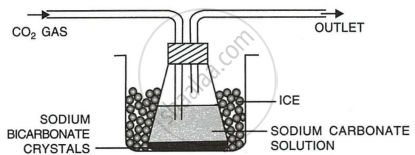
By passing carbon dioxide gas into a cold solution of sodium.
Reaction:
\[\ce{Na2CO3 + CO2 + H2O -> 2NaHCO3}\]
Procedure:
- Dissolve nearly 5 grams of anhydrous sodium carbonate in about 25 cm3 of distilled water in a flask.
- Cool the solution by keeping the flask in a freezing mixture.
- Pass carbon dioxide gas in the solution.
- Crystals of sodium bicarbonate will precipitate out after sometime.
- Filter the crystals and dry it in the folds of filter paper.
shaalaa.com
Laboratory Preparation of Some Salts - Laboratory Preparation of an Acid Salt Sodium Bicarbonate
Is there an error in this question or solution?
