Advertisements
Advertisements
Question
Consider the following compounds:
(i) p-methyl aniline
(ii) N, N-dimethylaniline
(iii) N-ethyl aniline
(iv) N-ethyl-N-methyl aniline
The compounds which do not form diazonium salt with ice-colds NaNO2 and HCl are:
Options
(i), (ii) and (iii)
(ii), (iii) and (iv)
(i), (iii) and (iv)
(ii) and (iv)
MCQ
Solution
(ii), (iii) and (iv)
Explanation:
Diazonium salt is formed only when the –NH2 group is directly bonded to the benzene ring.
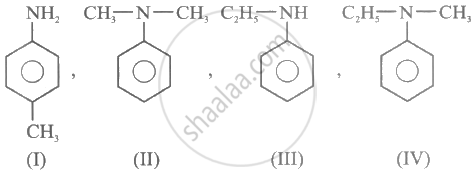
As a result, compounds (ii), (iii) and (iv) do not form diazonium salt.
shaalaa.com
Is there an error in this question or solution?
