Advertisements
Advertisements
Question
Identify the X and Y in the following reactions.
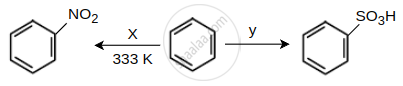
Options
\[\ce{X = Conc. HNO3, Y = H2SO4(SO3)}\]
\[\ce{X = Conc. HNO3 + Conc. H2SO4, Y = H2SO4(SO3)}\]
\[\ce{X = NaNO2/HCl, Y = H2SO4}\]
\[\ce{X = Dil{.} HNO3, Y = SO3/H2O2}\]
MCQ
Solution
\[\ce{X = Conc. HNO3 + Conc. H2SO4, Y = H2SO4(SO3)}\]
Explanation:
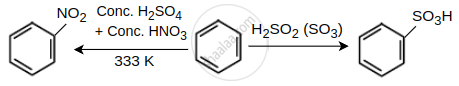
At 333 K, benzene produces nitrobenzene when it reacts with concentrated H2SO4 and concentrated HNO3, whereas it produces benzene sulphonic acid when it reacts with SO3 and concentrated H2SO4 (fumes) (Y).
shaalaa.com
Aromatic Hydrocarbons
Is there an error in this question or solution?
