Advertisements
Advertisements
Question
If λa, λb and λc represent the Kα, Kβ and Lα transition wavelengths in a hydrogen atom, respectively. Then which of the following is correct?
Options
`1/lambda_"A"+1/lambda_"B"=1/lambda_"C"`
`1/lambda_"C"-1/lambda_"B"=1/lambda_"A"`
`1/lambda_"B"+1/lambda_"C"=1/lambda_"A"`
`1/lambda_"A"+1/lambda_"C"=1/lambda_"B"`
MCQ
Solution
`bb(1/lambda_"A"+1/lambda_"C"=1/lambda_"B")`
Explanation:
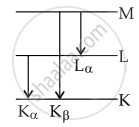
`"E"_("K"_alpha) + "E"_("L"_alpha)` = EKB
⇒ `1/lambda_"A"+1/lambda_"C"=1/lambda_"B"`
shaalaa.com
Is there an error in this question or solution?
