Advertisements
Advertisements
Question
If AB4 molecule is a polar molecule, a possible geometry of AB4 is ______.
Options
Square pyramidal
Rectangular planar
Square planar
Tetrahedral
MCQ
Fill in the Blanks
Solution
If AB4 molecule is a polar molecule, a possible geometry of AB4 is Square pyramidal.
Explanation:
For AB4 molecule the possible geometry are:
| S.No. | Geometry | H.O | b.p | 1.p | Nature of molecule |
| 1 | Tetrahedral | sp3 | 4 | 0 | Non-polar |
| 2 | Rectangular planar | sp3d | 4 | 1 | Non-polar |
| 3 | Square planar | sp3d2 | 4 | 2 | Non-polar |
Square pyramidal is polar because the structure is
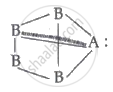
The dipole moment of A is never cancelled by others.
shaalaa.com
Is there an error in this question or solution?
