Advertisements
Advertisements
Question
In a body centred and face centred arrangement of atoms of an element, what will be the number of atoms present in respective unit cells? Justify your answer with a calculation.
Solution
In bed unit cell, the atoms are present only at the comers of a cube.
Total number of atoms in one unit cell = `8 xx 1/8 = 1`
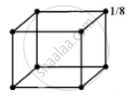
In face centred cubic unit cell,
The atoms are present at the comers as well as at the centres of six faces of a cube.
Total number of atoms in one unit cell
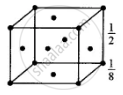
`= 8 xx 1/8 + 6 xx 1/2`
= 1 + 3 = 4
APPEARS IN
RELATED QUESTIONS
A10% aqueous solution of cane sugar (mol. wt. 342) is isotonic with 1.754% aqueous solution of urea. Find the molecular mass of urea.
A compound AB has a simple cubic structure and has molecular mass 99. its density is 3.4 g cm-3. What will be the edge length of the unit cell?
An equimolar solution of non-volatile solutes A and B shows a depression in freezing point in the ratio of 2:1. If A remains in its normal state in the solution, the state of B in the solution will be ______.
