Advertisements
Advertisements
Question
In a hydrogen atom, an electron of charge e revolves in a orbit of radius r with speed v. Then, the magnetic moment associated with electron is ______.
Options
`"evr"/2`
2 evr
evr
`"evr"/3`
Solution
In a hydrogen atom, an electron of charge e revolves in a orbit of radius r with speed v. Then, magnetic moment associated with electron is `underline("evr"/2)`.
Explanation:
Magnetic moment of revolving electron,
μ = i × A
where, i = current and A = area,
`therefore mu_"e" = "e"/"T" xx pi"r"^2` (As current, i = `"charge"/"time" = "e"/"T"`)
where, T = time period of revolution and r = radius of orbit.
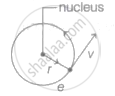
`mu_"e" = "e"/(2pi"r") xx "v" xx pi"r"^2 ....(because "Time period" ("T") = "distance"/"speed")`
where, v = velocity of revolving electron
`=> mu_"e" = "evr"/2`
Hence, the magnetic moment of revolving electron is `"evr"/2`.
