Advertisements
Advertisements
Question
In carius method of estimation of halogen 0.172 g of an organic compound showed presence of 0.08 g of bromine. Which of these is the correct structure of the compound?
Options
CH3–Br
CH3 CH2–Br
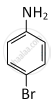
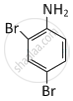
MCQ
Solution

Explanation:
No. of mole of bromine = `0.08/80` = 0.001 mole = 10−3 mole.
Let the molecular weight of the compound is M.
When 10−3 mole of bromine is present, weight of organic compound is 0.172 g.
When 1 mole of bromine is present, weight of organic compound = `0.172/10^-3` g mol−1
∴ M = `0.172/10^-3` = 172 g mol−1
Therefore the compound is

As its molecular weight = 80 + 72 + 14 + 6 = 172
shaalaa.com
Quantitative Analysis of Halogens
Is there an error in this question or solution?
