Advertisements
Advertisements
Question
In \[\ce{Cu^{2+} + Ag -> Cu + Ag^+}\], oxidation half-reaction is:
Options
\[\ce{Cu^{2+} -> Cu}\]
\[\ce{Ag -> Ag^+}\]
\[\ce{Cu -> Cu^{2+}}\]
All of these
MCQ
Solution
\[\ce{Ag -> Ag^+}\]
Explanation:
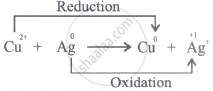
Thus, the oxidation-half reaction is \[\ce{Ag -> Ag^+}\]
shaalaa.com
Balancing Redox Reactions in Terms of Loss and Gain of Electrons
Is there an error in this question or solution?
