Advertisements
Advertisements
Question
In gas phase H-O-O-H bond angle in H2O2 is ______.
Options
94.8°
111.5°
98.4°
147.5°
MCQ
Solution
In gas phase H-O-O-H bond angle in H2O2 is 94.8°.
Explanation:
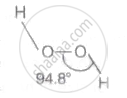
There are lone pairs on the surface of the gas phase. oxygen and to reduce the number of lone-pair-lone pairs. The binding angle was lowered from 101.9° to 101.9° due to repulsion. 94.8° (crystalline phase) (gas phase).
shaalaa.com
Parameters of Covalent Bond
Is there an error in this question or solution?
