Advertisements
Advertisements
Question
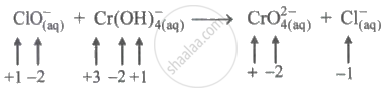
In the following reaction, the oxidation number of Cr changes.
Options
+6 to +3
+2 to +4
+4 to +2
+3 to +6
MCQ
Solution
+3 to +6
Explanation:
shaalaa.com
Oxidation Number
Is there an error in this question or solution?
