Advertisements
Advertisements
Question
In the following reaction, the reason for why meta-nitro product also formed is:

Options
Formation of anilinium ion
−NO2 substitution always takes place at meta-position.
low temperature
−NH2 group is highly meta-directive.
MCQ
Solution
Formation of anilinium ion
Explanation:
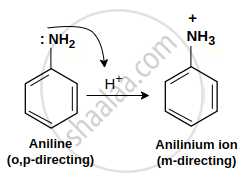
During aniline nitration in the presence of an acid, the −NH2 group is protonated and converted to \[\ce{NH^+3}\] (anilinium ion being electron withdrawing group gives meta product).
shaalaa.com
Is there an error in this question or solution?
