Advertisements
Advertisements
Question
In the nth orbit, the energy of an electron `"E"_"n"= -13.6/"n"^2"eV"` for hydrogen atom. The energy required to take the electron from first orbit to second orbit will be ____________.
Options
10.2 eV
12.1 eV
13.6 eV
3.4 eV
MCQ
Fill in the Blanks
Solution
In the nth orbit, the energy of an electron `"E"_"n"= -13.6/"n"^2"eV"` for hydrogen atom. The energy required to take the electron from first orbit to second orbit will be 10.2 eV.
Explanation:
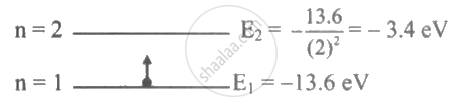
`therefore E_1→_2 = -3.4 - (13.6) = + 10.2 "eV"`
shaalaa.com
Bohr’s Atomic Model
Is there an error in this question or solution?
