Advertisements
Advertisements
Question
In the reaction,
\[\ce{CH3COOH ->[SOCl2] X ->[Sodium salt of carboxylic acid] Y}\].
The compound Y was found to be a mixed acid anhydride. Thus, the sodium salt of carboxylic acid used CANNOT be ____________.
Options
sodium propionate
sodium acetate
sodium butyrate
sodium valerate
Solution
In the reaction,
\[\ce{CH3COOH ->[SOCl2] X ->[Sodium salt of carboxylic acid] Y}\].
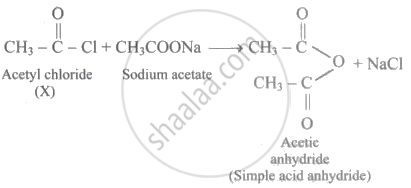
The compound Y was found to be a mixed acid anhydride. Thus, the sodium salt of carboxylic acid used CANNOT be sodium acetate.
Explanation:
\[\begin{array}{cc}
\phantom{}\ce{O}\phantom{.............................}\ce{O}\phantom{.....................}\\
\phantom{}||\phantom{.............................}||\phantom{.....................}\\
\ce{\underset{\text{Acetic acid}}{CH3-C-OH} + SOCl2 ->[\Delta][Pyridine] \underset{\text{Acetyl chloride (X)}}{CH3-C-Cl} + SO2 ^ + HCl ^}
\end{array}\]
Because a mixed acid anhydride was produced as a result of the additional reaction of X (i.e., acetyl chloride) with the sodium salt of carboxylic acid. As a result, sodium acetate cannot be utilised as a salt. This is because the reaction would yield simple acid anhydride (viz., acetic anhydride) as follows: