Advertisements
Advertisements
Question
In the table below, H does not represent hydrogen. Some elements are given in their own symbol and position in the periodic table while others are shown with a letter.
| IA | IIA | IIIA | IVA | VA | VIA | VIIA | O |
| 1 | 2 | 13 | 14 | 15 | 16 | 17 | 18 |
| Li | D | O | J | Ne | |||
| A | Mg | E | Si | H | K | ||
| A | C | F | G | L |
Select from the table.
- Which is most electronegative.
- How many valence electrons are present in G.
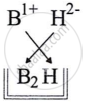
- Write the formula of the compound between B and H.
- In the compound between F and J what type of bond will be formed.
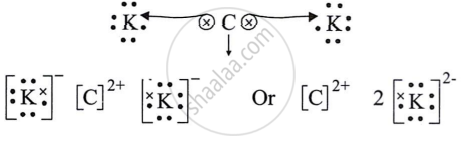
- Draw the electron dot structure for the compound formed between C and K.
Solution
(i) J is the most electronegative.
(ii) G has group 15 or VA, indicating it will have 5 electrons in its outermost shell.
(iii) B has one valence electron and its valency is +1. It is represented as B1+.
H has 6 valence electrons, resulting in a valency of −2 (represented as H2−).
Chemical formula=
(iv) F belongs to the 14th group, also known as IVA group. Four valence electrons indicate a covalent bond where electrons are covalent.
(v) C and K have two and seven valence electrons, respectively. To complete an octet, K requires one electron, while C requires two electrons to lose.