Advertisements
Advertisements
Question
In which of the following reaction, H2O2 is behaving as a reducing agent:
Options
\[\ce{C6H6 + H2O2 -> C6H5OH + H2O}\]
\[\ce{PbS + 4H2O2 -> PbSO4 + 4H2O}\]
\[\ce{2I^- + 2H^+ + H2O2 -> I2 + 2H2O}\]
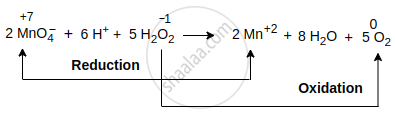
\[\ce{2MnO^-_4 + 6H^+ + 5H2O2 -> 2Mn^{2+} + 5O2 + 8H2O}\]
MCQ
Solution
\[\ce{2MnO^-_4 + 6H^+ + 5H2O2 -> 2Mn^{2+} + 5O2 + 8H2O}\]
Explanation:

shaalaa.com
Classical Idea of Redox Reactions - Oxidation and Reduction Reactions
Is there an error in this question or solution?
