Advertisements
Advertisements
Question
Indicate the number of unpaired electrons in Kr.
Solution
Krypton (Kr):
Atomic number = 36
The electronic configuration is:
1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6
The orbital picture of krypton is:
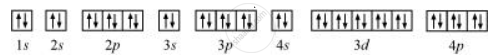
Since all orbitals are fully occupied, there are no unpaired electrons in krypton.
APPEARS IN
RELATED QUESTIONS
Which atom is indicated by the following configuration?
[He] 2s1
Which atom is indicated by the following configuration?
[Ne] 3s2 3p3
Which atom is indicated by the following configuration?
[Ar] 4s2 3d1.
An atomic orbital has n = 3. What are the possible values of l and ml?
List the quantum numbers (ml and l) of electrons for 3d orbital.
Which of the following orbitals are possible?
1p, 2s, 2p and 3f
Indicate the number of unpaired electrons in a P.
Indicate the number of unpaired electrons in Si.
Indicate the number of unpaired electrons in Cr.
Indicate the number of unpaired electrons in Fe.
Show the distribution of electrons in oxygen atom (atomic number 8) using orbital diagram.
The atomic number of elements is 17. The number of orbitals containing electron pairs in its valence shell is ______.
Which of the following statements is correct?
(a) The electronic configuration of Cr is (Ar) 3d5 4s1.
(b) The magnetic quantum number may have a negative value.
(c) In the ground state of an atom, the orbitals are filled in order of their increasing energies.
(d) The total number of nodes are given by n − 2.
Choose the most appropriate answer from the options given below:
The number of 4f electron in the ground state electronic configuration Gd2+ is ______. (Atomic number of Gd = 64)
The correct set of four quantum numbers for the valence electrons of the rubidium atom (Z = 37) is ______.
