Advertisements
Advertisements
Question
Magnesium ribbon on burning in air produces
Options
magnesium oxide, water and light
magnesium oxide and heat
magnesium oxide, heat and light
magnesium oxide, water and heat
Solution
magnesium oxide, heat and light
Explanation -
Magnesium ribbon on burning in air produces magnesium oxide, heat and light, Magnesium ribbon is a very reactive metal.
\[\ce{Magnesium + \underset{(From air)}{Oxygen}\overset{Combustion}{->} Magnesium oxide + Heat + Light}\]
APPEARS IN
RELATED QUESTIONS
Make a labelled diagram of a candle flame.
True or False – If False give the correct answer
All fuels form flame.
Picture based question
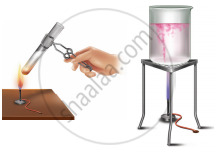
Arul and Aakash were doing an experiment in which water was to be heated in a beaker. Arul kept the beaker near the wick in the yellow part of candle flame. Aakash kept the beaker in the outer most part to the flame. Whose water will get heated in a shorter time?
The substance that does not burn with flame is
The substances which gave very ______ ignition temperature and can easily catch fire with a flame are called ______ substances.
Match the following for the flame of a candle.
| Column A |
Column B (zone) |
Column C (colour) |
| (a) hottest part | (iii) outer zone of complete combustion | (x) blue |
| (b) moderately hot | (ii) middle zone of partial combustion | (z) yellow |
| (c) least hot | (i) innermost zone of complete combustion | (y) black |
If you hold a piece of iron wire with a pair of tongs inside a candle flame or a Bunsen burner flame, what will you observe? Will it produce a flame?
Substances which vapourise during burning produce ______.
Write a note on flame.
Explain the structure of a candle flame.
