Advertisements
Advertisements
Question
Match the terms in List I with the correct answers in List II.
| List I | List II |
| 1. Solubility in a given solvent decreases with an increase in temperature. | A: Na2SO4.10H2O |
| 2. A white efflorescent crystal | B: NaOH |
| 3. A hygroscopic dehydrating agent | C: Ca(HCO3)2 |
| 4. A deliquescent alkali | D: conc. H2SO4 |
| 5. A substance present in temporary hard water | E: CaSO4 |
Solution
| List I | List II |
| 1. Solubility in a given solvent decreases with an increase in temperature. | E: CaSO4 |
| 2. A white efflorescent crystal | A: Na2SO4.10H2O |
| 3. A hygroscopic dehydrating agent | D: conc. H2SO4 |
| 4. A deliquescent alkali | B: NaOH |
| 5. A substance present in temporary hard water | C: Ca(HCO3)2 |
APPEARS IN
RELATED QUESTIONS
Answer the following question:
Why is acid added to water before electrolysis ?
How is rain produced?
State whether the given statement are true or false. If true, write the reason.
Ganga−Brahmaputra plain of India is an overpopulated region.
Why is water considered a compound?
Identify A, B, C and D; first one is done for you.
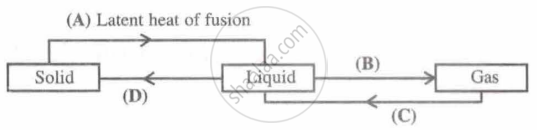
Explain why:
Air dissolved in water contains a higher proportion of oxygen.
Give a balanced equation for the following conversion:
Calcium sulphate in permanent hard water to calcium carbonate using sodium carbonate.
Explain the following observation:
Washing-soda become coated with a white powder when left exposed to the atmosphere.
Select the correct word from the words in option to complete the sentence:
If pressure on the surface of water increases its boiling point ____ and freezing point ____.
Look at the map of Tamilnadu showing annual rainfall and answer the questions given below

- Identify the districts that get only low annual rainfall in Tamilnadu.
- Identify the districts that get a medium annual rainfall in Tamilnadu.
- State the districts that enjoy high annual rainfall in Tamilnadu.
