Advertisements
Advertisements
Question
Molecular compounds are usually formed by the combination between :
Options
a metal and a non-metal
two different non-metals
two different metals
any two gaseous elements
Solution
two different non-metals
Molecular compounds are formed by the combination of two different non-metals.
APPEARS IN
RELATED QUESTIONS
Give the name of the element present in the following compound:
Quick lime
Define: Mass number :
What is the difference between the molecule of an element and the molecule of a compound ? Give one
example of each.
An element X forms the following compounds with hydrogen, carbon and oxygen :
H2X, CX2, XO2, XO3
State the three valencies of element X which are illustrated by these compounds.
Explain the term – molecules. Give three examples of atoms of the same element forming a molecule. State the atomicity of the same.
Give one example of a triatomic molecule
State the main points of the contradiction of Dalton’s atomic theory by the Modern Atomic Theory.
State the atomicity of the following:
Oxygen
State the atomicity of the following:
Chlorine
Classify the following molecules as the molecules of element or compound
| 1. |  |
|
| 2. |  |
|
| 3. | 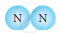 |
|
| 4. | 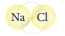 |
