Advertisements
Advertisements
Question
Name a compound and draw the figure:
Cyclic compound with a single bond.
Solution
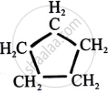
Cyclopentane
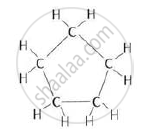
Or
APPEARS IN
RELATED QUESTIONS
Identify the term or substance based on the descriptions given below:
Hydrocarbon containing a triple bond used for welding purposes.
Complete and balance the following equation. State the condition wherever necessary.
\[\begin{array}{cc}
\ce{CH}\phantom{..............................}\\
\ce{|||\phantom{..}+ H2 -> \underline{}\underline{}\underline{}\underline{} + H2 -> \underline{}\underline{}\underline{}\underline{}}\\
\ce{CH}\phantom{..............................}
\end{array}\]
Give the names and structural formula of saturated hydrocarbon ?
Which type of reaction will they undrgo?
Give the name and structural formula of unsaturated hydrocarbon ?
Which type of reaction will they undrgo?
Explain the following : Catenation
Give at least one example in case to show the structure of isomers of single-bond compounds.
Draw the structural formula for the following compound:
vinegar
Fill in the blanks with the correct words from the brackets:
Alkenes are the (i) ______ (analogous/homologous) series of (ii) ______ (saturated/unsaturated) hydrocarbons. They differ from alkanes due to presence of (c) ______ (double/single) bonds. Alkenes mainly undergo (iv) ______ (addition/substitution) reactions.
Give one word for the following statement:
Organic compounds having the same molecular formula but different Structural formula.
The structures of four hydrocarbons are shown below:
| \[\begin{array}{cc} \phantom{.....}\ce{CH3}\\ \phantom{...}|\\ \ce{H3C - C - H}\\ \phantom{...}|\\ \phantom{.....}\ce{CH3} \end{array}\] |
\[\begin{array}{cc} \ce{CH3}\phantom{.....}\\ |\phantom{.......}\\ \ce{C = CH2}\\ |\phantom{.......}\\ \ce{CH3}\phantom{....} \end{array}\] |
\[\begin{array}{cc} \ce{H}\phantom{.....}\\ |\phantom{.....}\\ \ce{H3C - C - C - CH2}\\ |\phantom{....}|\\ \ce{H\phantom{...}H} \end{array}\] |
\[\begin{array}{cc} \phantom{...}\ce{CH3}\\ |\\ \ce{H3C - C = CH2} \end{array}\] |
How many isomers of butene are there?
