Advertisements
Advertisements
Question
Number of lone pair(s) of electrons on central atom and the shape of BrF3 molecule respectively are ______.
Options
0, triangular planar
1 pyramidal
2, bent- T-shape
1, bent T-shape
MCQ
Fill in the Blanks
Solution
Number of lone pair(s) of electrons on central atom and the shape of BrF3 molecule respectively are 2, bent- T-shape.
Explanation:
In BrF3 molecule, there are 2 lone pairs and 3 bond pairs of electrons. The central Br atom undergoes sp3d hybridisation. The expected geometry of BrF3 is trigonal bipyramidal and actual geometry is T-shaped since 2 of the orbitals are occupied by lone pairs.
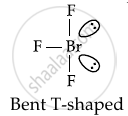
shaalaa.com
Is there an error in this question or solution?
