Advertisements
Advertisements
Question
Out of C and CO, which is a better reducing agent at 673 K?
Solution 1
At 673 K, the value of `triangleG_(CO,CO_2)` is less than that of `triangleG_(("C,CO"))` Therefore, CO can be oxidised more easily to CO2 than C to CO. Hence, CO is a better reducing ag ent than C at 673 K.
Solution 2
This can be explained thermodynamically, taking entropy and free energy changes into account.
`C(s) + O_2(g) -> CO_2(g)`
`2C(s) + O_2(g) -> 2CO (g)`
Case i: Volume of CO2 produced = Volume of O2 used.
`:.triangleS` is very small and `triangleG` does not change with temperature.
:.Plot of `triangleG` Vs T is almost hrizontal
Case ii: Volume of CO produced = 2 x Volume of O2 used
`:.triangleS` is positive and hence `triangleG` beccomes increasingly negative as the temperature increases.
:.Plot of `triangle ""^@G` Vs T slopes downwards.
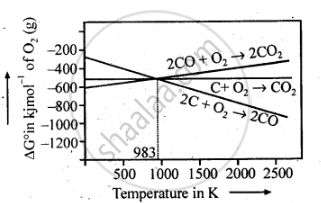
As can be seen from ΔG° Vs T plot (Ellingham diagram), lines for the reactions, C ——–> C02 and C ——–> CO cross at 983 K. Below 983 K, the reaction (a) is energetically more favourable but above 673 K, reaction (b) is favourable and preferred. Thus, below 673 K both C and CO can act as a reducing agent but since CO can be more easily oxidised to C02 than C to C02 , therefore, below 673 K, CO is more effective reducing agent than carbon
APPEARS IN
RELATED QUESTIONS
Giving examples, differentiate between ‘roasting’ and ‘calcination’.
Why copper matte is put in silica lined converter?
Out of C and CO, which is a better reducing agent for ZnO ?
Which one of the following is not feasible
Flux is a substance which is used to convert
Zinc is obtained from ZnO by ____________.
What are the various steps involved in the extraction of pure metals from their ores?
What is the role of Limestone in the extraction of iron from its oxide Fe2O3?
Explain the following term with a suitable example.
Gangue
Explain the following term with a suitable example.
Slag
Which reagents are required for one step conversion of chlorobenzene to toluene?
The reaction of toluene with Cl2 in presence of FeCl3 gives ‘X’ while the of toluene with Cl2 in presence of light gives ‘Y’. Thus ‘X’ and ‘Y’are ______.
At the temperature corresponding to which of the points in figure, FeO will be reduced to Fe by coupling the reaction \[\ce{2FeO -> 2Fe + O2}\] with all of the following reactions?
(a) \[\ce{C + O2 -> CO2}\]
(b) \[\ce{2C + O2 -> 2CO}\]
(c) \[\ce{2CO + O2 -> 2CO2}\]
(i) Point A
(ii) Point B
(iii) Point D
(iv) Point E
CN– solution is used in the extraction of which metal?
Colemanite is:-
The process that involves the removal of sulphur from the ores is ______.
