Advertisements
Advertisements
Question
Plot a graph showing variation of de Broglie wavelength λ versus `1/sqrtV` , where V is accelerating potential for two particles A and B, carrying the same charge but different masses m1, m2 (m1 > m2). Which one of the two represents a particle of smaller mass and why?
Solution
Given: Charge (q) is the same but masses of both the particles are different (m1 > m2).
The de Brogile wavelength given by
`lambda = h/sqrt(2mqV)`
The slope of the graph λ versus `1/sqrtV `is `h/sqrt(2mq)`
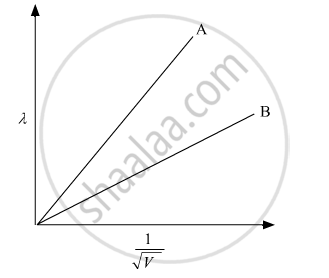
The slope of the smaller mass is larger; therefore, plot A in the above graph is for mass m2
RELATED QUESTIONS
A proton and an α -particle are accelerated through the same potential difference. Which one of the two has greater de-Broglie wavelength Justify your answer.
A proton and a deuteron are accelerated through the same accelerating potential. Which one of the two has the greater value of de-Broglie wavelength associated with it, and Give reasons to justify your answer.
State de Broglie hypothesis
A proton and an alpha particle are accelerated through the same potential. Which one of the two has (i) greater value of de−Broglie wavelength associated with it and (ii) less kinetic energy. Give reasons to justify your answer.
An electron of energy 150 eV has wavelength of 10-10m. The wavelength of a 0.60 keV electron is?
Gas exerts pressure on the walls of the container because :
For an electron accelerated from rest through a potential V, the de Broglie wavelength associated will be:
For production of characteristic of X-rays the electron trans mission is
The kinetic energy of electron in (electron volt) moving with the velocity of 4 × 106 m/s will be
A neutron beam of energy E scatters from atoms on a surface with a spacing d = 0.1 nm. The first maximum of intensity in the reflected beam occurs at θ = 30°. What is the kinetic energy E of the beam in eV?
