Advertisements
Advertisements
Question
Pratyush took sulphur powder on a spatula and heated it. He collected the gas evolved by inverting a test tube over it, as shown in the figure below.
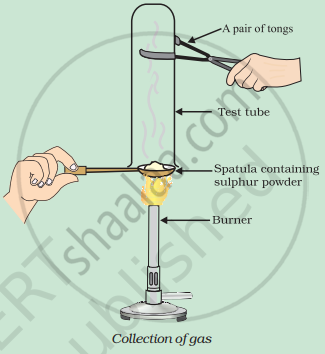
- What will be the action of gas on
- dry litmus paper?
- moist litmus paper?
- Write a balanced chemical equation for the reaction taking place.
Answer in Brief
Solution
-
- There will be no action on dry litmus paper.
- Litmus paper turns red because sulphur is a nonmetal, and the oxides of nonmetals are acidic. On prolonged contact with the gas, the litmus paper becomes decolourised.
- \[\ce{S_{(s)} + O2_{(g)} ->[\Delta] SO2_{(g)}}\]
\[\ce{SO2_{(g)} + H2O_{(l)} -> \underset{(turns moist blue litmus red)}{H2SO3_{(aq)}}}\]
shaalaa.com
Is there an error in this question or solution?
