Advertisements
Advertisements
Question
Predict conditions under which Al might be expected to reduce MgO.
Solution 1
Above 1350°C, the standard Gibbs free energy of formation of Al2O3 from Al is less than that of MgO from Mg. Therefore, above 1350°C, Al can reduce MgO.
Solution 2
The equations for the formation of the two oxides are
`4/3 Al(s) + O_2(s) -> 2/3 Al_2O_3(s)`
`2Mg(s)+O_2(s) -> 2MgO(s)`
`Double arrow`
If we look at the plots for the formation of the two oxides of the Ellingham diagram, we find that they intersect at certain point. The corresponding value of ΔG° becomes zero for the reduction of MgO by Al metal.
2MgO(s) + 4/3Al(s) ⇌ 2Mg(s) + 2/3Al2O3(s)
This means that the reduction of MgO by Al metal can occur below this temperature. Aluminium (Al) metal can reduce MgO to Mg above this temperature because Δ°G for Al2O3 is less as compared to that of MgO.
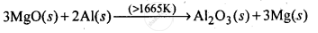
APPEARS IN
RELATED QUESTIONS
What are Ellingham's diagrams? Write 2 features of it.
Why is zinc not extracted from zinc oxide through reduction using CO?
The value of `triangle_tG^theta` for formation of Cr2O3 is − 540 kJmol−1 and that of Al2 O3 is − 827 kJmol−1. Is the reduction of Cr2O3 possible with Al?
Write features of Ellingham's diagrams
Silica is a/an _______.
Given below are two statements.
Statements I: The choice of reducing agents for metal extraction can be made by using Ellingham diagram, a plot of ΔG vs temperature.
Statements II: The value of ΔS increases from left to right in Ellingham diagram. In the light of above statements, choose the most appropriate from the options given below:
With respect to an ore, Ellingham diagram helps to predict the feasibility of its.
In blast furnace, the hearth is lined with ______.
The o, p-directing but deactivating group is ______.
Assertion A: Magnesium can reduce Al2O3 at a temperature below 1350°C, while above 1350°C aluminium can reduce MgO.
Reason R: The melting and boiling points of magnesium are lower than those of aluminium.
In light of the above statements. choose most appropriate answer from the options given below:
An Ellingham diagram provides information about ______.
