Advertisements
Advertisements
Question
Since metal carbonate and metal bicarbonate are basic, they react with acids to give salt and water with the liberation of ______.
Options
NO2
SO2
CO2
Solution
Since metal carbonate and metal bicarbonate are basic, they react with acids to give salt and water with the liberation of CO2
APPEARS IN
RELATED QUESTIONS
When a solution is added to vanilla extract, then the characteristic smell of vanilla cannot be detected. State whether the given solution is an acid or a base.
Name a sodium compound which is a constituent of many dry soap powders.
Water is never added to acid in order to dilute it why?
Define the term ‘basicity’ of an acid.
Explian the following:
Dil. HCl acid is stronger than highly concentrated acetic acid.
Write balanced equation to satisfy the following statement:
\[\ce{Acid + Carbonate or bicarbonate -> Salt + water + Carbon dioxide}\]
Give the appropriate term defined by the statements given below :
The substance that releases hydronium ion as the only positive ion when dissolved in water.
What do you understand by the strength of an acid? On what factors does the strength of an acid depend?
Fill in the crossword given in Figure 5.2 with the help of the clues provided.
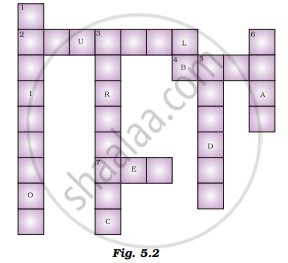
Across
(2) The solution which does not change the colour of either red or blue litmus.
(4) Phenolphthalein gives pink colour in this type of solution.
(7) Colour of blue litmus in lemon juice.
Down
(1) It is used to test whether a substance is acidic or basic.
(3) It is a natural indicator and gives pink colour to the basic solution.
(5) Nature of ant’s sting.
(6) It is responsible for the increase in temperature during a neutralisation reaction.
Why does blue color copper sulphate become white on heating?
