Advertisements
Advertisements
Question
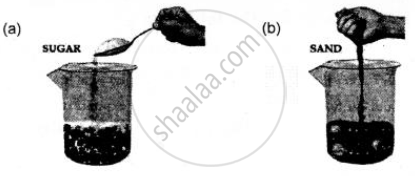
State what would you observe if
(a) sugar is added to pebbles take in a plastic beaker
(b) sand is added to glass balls in a beaker. What would you conclude from this imaginative demonstration?
Solution
Experiment: Add sugar to pebbles taken in a plastic beaker or sand to glass balls in a beaker.
Observation: The sugar or the sand goes into the space between the pebbles & the glass balls respectively.
Conclusion: An imaginative demonstration to show that intermolecular spaces between particles are occupied easily.
RELATED QUESTIONS
Ice melts at 100°C.
Evaporation of a liquid causes cooling.
Evaporation takes place over the entire mass of the liquid.
Write true or false for the following statement
In a gas, the molecules can move anywhere in space.
How a liquid changes into its gaseous state? Explain?
Fill in the blank
Liquids have no definite ...........
Fill in the blank
When a gas is cooled, its molecules .............. energy.
Write whether the following statement is true or false.
Intermolecular force of attraction is greater in gases than in liquids.
For the following statement, say whether it describes a solid, a liquid, or a gas.
Particles move about very quickly.
What is meant by the inter-molecular forces of attraction ?
