Advertisements
Advertisements
Question
Given below are two statements.
- Statement I: In CuSO4 · 5H2O, Cu-O bonds are present.
- Statement II: In CuSO4 · 5H2O, ligands coordinating with Cu(II) ion are O- and S based ligands.
In light of the above statement, choose the correct answer from the options given below.
Options
Both Statement I and Statement II are correct.
Both Statement I and Statement II are incorrect.
Statement I is correct but Statement II is incorrect.
Statement I is incorrect but Statement II is correct.
MCQ
Solution
Statement I is correct but Statement II is incorrect.
Explanation:
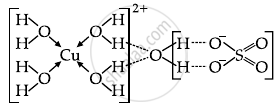
A covalent bond is formed between sulphur and oxygen as they both are non-metals and the bond between copper and sulphate is ionic one is metal and the other is non-metal. A compound called hydrated copper sulphate is bound to water molecules, which serve as ligands to establish coordinate bonds.
shaalaa.com
Is there an error in this question or solution?
