Advertisements
Advertisements
Question
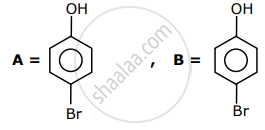
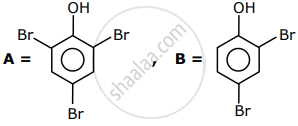
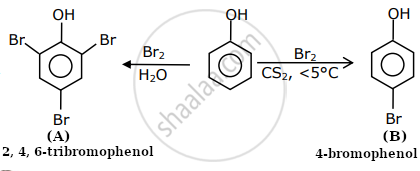
The correct options for the products A and B of the following reactions are:
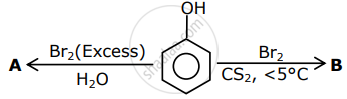
Options
MCQ
Solution
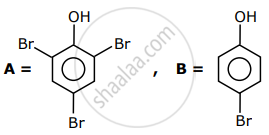
Explanation:
Excessive bromination occurs when polar solvent is present, whereas mono-bromination on phenol occurs when a non-polar solvent is present. In the presence of polar solvent, phenol is present as phenoxide, which increases the rate of EAS, but in the presence of non-polar solvent, it is present as phenol alone, which results in mono-brominated phenol.
shaalaa.com
Alcohols, Phenols and Ethers
Is there an error in this question or solution?