Advertisements
Advertisements
Question
The dipole moments of the given molecules are such that ______.
Options
BF3 > NF3 > NH3
NF3 > BF3 > NH3
NH3 > NF3 > BF3
NH3 > BF3 > NF3
MCQ
Fill in the Blanks
Solution
The dipole moments of the given molecules are such that NH3 > NF3 > BF3.
Explanation:

BF3 is symmetrical molecular, `μ_("BF"_3)` = 0
`mu_"r"` is neutralized by the moment of third bond B 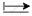 F. Therefore µ = 0
F. Therefore µ = 0
`therefore mu_("NH"_3) > mu_("NF"_3) > mu_("BF"_3)`
or NH3 > NF3 > BF3
shaalaa.com
Is there an error in this question or solution?
