Advertisements
Advertisements
Question
The energy levels with transitions for the atom are shown. The transitions corresponding to emission of radiation of maximum and minimum wavelength are respectively.
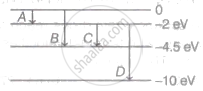
Options
B, C
A, C
C, D
A, D
MCQ
Solution
A, D
Explanation:
As, we know that the difference of energy between two level is equal to the energy of photon.
`Delta"E" = "hv" = "hc"/lambda => lambda = "hc"/(Delta "E")`
or `lambda prop 1/(Delta "E")`
The value of ΔE is minimum for A, so radiation corresponding to A have maximum wavelength and value of Δ E is maximum for D, so radiation corresponding to D have minimum wavelength.
shaalaa.com
Emission of Heat Radiation
Is there an error in this question or solution?
