Advertisements
Advertisements
Question
The integrated rate equation is Rt = log C0 – log Ct, then the straight-line graph is obtained by plotting.
Options
Time vs loge
`1/"time"` vs Ct
Time vs Ct
`1/"time"` vs `1/(C_t)`
MCQ
Solution
Time vs loge
Explanation:
For first-order reaction,
Integrated rate equation, `kt = log C_0 - log C_t`
The straight-line graph is obtained by plotting
`log C_t` / versus time.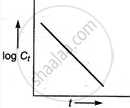
shaalaa.com
Is there an error in this question or solution?
