Advertisements
Advertisements
Question
The number of unpaired electrons in the complex ion [NiCl4]2− is ____________.
Options
0
1
2
4
MCQ
Fill in the Blanks
Solution
The number of unpaired electrons in the complex ion [NiCl4]2− is 2.
Explanation:
Configuration after [NiCl4]2− complex formation is:
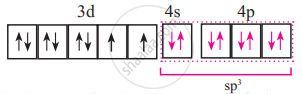
It has 2 unpaired electrons.
shaalaa.com
Theories of Bonding in Complexes
Is there an error in this question or solution?
